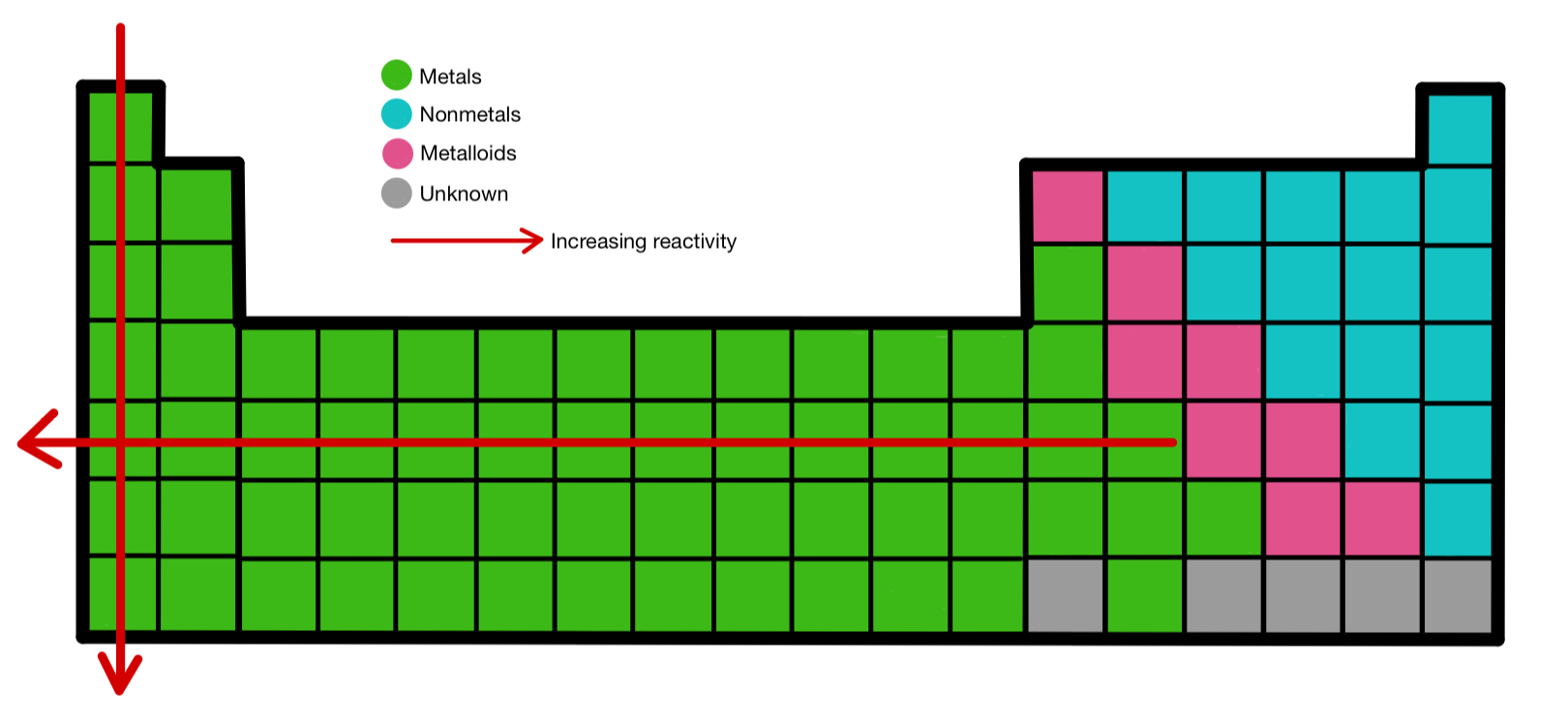
WebThe reactivity of Group 1 elements increases as you go down the group. What is the trend in reactivity on the period table? Socratic Trump arraignment: Judge bans use of digital devices, allows … In an order tonight, Judge Juan Merchan said no cellphones, laptops or broadcast equipment can be used in the courtroom during Trump’s hearing tomorrow. Making predictions about elements - BBC Bitesize Whatever these metals react with, they have to form positive ions in the process, and so the lower the ionisation energy, the more easily those ions will form. You can explain the increase in reactivity of the Group 1 metals (Li, Na, K, Rb, Cs) as you go down the group in terms of the fall in ionisation energy. 
the attraction between the nucleus and outer electron gets weaker as you go down the group – so the electron is more easily lost. 
The reactivity of Group 1 elements increases as you go down the group because: the outer electron gets further from the nucleus as you go down the group.How is the periodic table …Ĭhemistry lessons 9-15 Flashcards Quizlet Science - Recall Questions (Y10T2W1) Flashcards Quizlet WebWhy does reactivity change as you go down Group 1? The shielding and atomic radius increase, making it easier to lose the outer shell electron. Group 1 metals are very reactive, and must be stored out of contact with air to prevent oxidation. (Physical_and_Theoretical_Chemistry)/Physical_Properties_of_Matter/Atomic_and_Molecular_Properties/Electron_Affinity In an order tonight, Judge Juan Merchan said no cellphones, laptops or broadcast equipment can be used in the courtroom during Trump’s … Why is Group 2 more reactive as you go down? This makes it easier to remove the electron and makes the atom more reactive. The attraction from the positive nucleus to the negative electron is less. Therefore, the outermost electron gets further from the nucleus. Group 1: Reactivity (GCSE Chemistry) - Study Mind WebAs you go down group 1, the number of electron shells increases – lithium has two, sodium has three etc. The electrons in the outer shell move further away from the nucleus as we go down the group and the … The reactivity of Group 7 elements decreases down the group.Why do group 1 and 2 metals get more reactive as you go down … Verdugo rips Jays Web WebCreated by MrAustin12 Terms in this set (20) As you go down group 1 Softness increases, density increases, melting point decreases, reactivity increases. The first impression that is sometimes given that the fall in reactivity is because the incoming electron is held less strongly as you go down the group and so the negative ion is less likely to form.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
